|
File: <braco1.ima.htm> [For educational purposes only] Terminology Glossary <Principal Natural
Enemy Groups > <Citations> |
Immature Stages
of Braconidae
Immature stages of Braconidae were discussed in
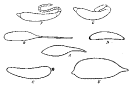
detail by Clausen (1940), as follows: The Egg.--The general form of the
eggs of the Braconidae is simple, ranging in outline from broadly oval to
almost cylindrical but frequently somewhat pear‑shaped, or elongate and
tapering at both ends and usually without a stalk or pedicel. The egg of Microbracon lendicivorus
(Fig. 13B) differs from those of other Vipioninae in having a slender
tapering stalk, slightly longer than the egg body, at what is presumably the
anterior end. In Dendrosoter
protuberans Nees (Fig. 13G),
the stalk is very broad and bent back upon the egg body in a characteristic
way, whereas, the stalk of Coeloides
subconcolor Russo (Fig. 13F)
is long and has a distinctly segmented appearance (Russo, 1938). Most of the species of Apanteles have a short peduncle
at the posterior end, and Opius
crawfordi Vier. has this
peduncle equal to or longer than the egg body. Other species of the latter
genus lack the peduncle entirely or have it in a very reduced form (Fig. 13D,
E). A thin, transparent membrane
possibly the exochorion envelops the egg of O. tryoni, which is broken during the period of incubation owing
to increase in size of the egg. This recalls a similar egg envelope found by
Dowden in Brachymeria compsilurae Cw. Please CLICK on
pictures to view details: The Euphorinae have the stalk at the posterior
end, and in some species of Perilitus
it is nearly as long as the main body. The same is true in a number of
species of Meteorinae. The egg of Alysia manducator (Fig. 13C) bears a pronounced buttonlike
tubercle at its larger, presumably anterior end. Although the evidence is not complete and there are several
apparent exceptions, it appears that tho stalk of the braconid egg, when
present, is usually situated at the posterior end. In no instance does it serve any definite purpose after
deposition. The eggs of Ascogaster, Phanerotoma,
and Chelonus, which are
dcposited in those of the host, are of minute size, measuring 0.2 mm. or less
in length. In all species of the
family the chorion is thin and transparent, and usually has no surface
sculpturing, though in Meteorus
versicolor and Microdus dimidiatus it bears minute hexagonal markings. First‑instar Larvae. --The
first‑instar larvae of the Braconidae represent a considerable variety
of forms, comprising the hymenopteriform, mandibulate, caudate, vesiculate
and polypodeiform. It is often
debatable; particularly in reference to this family, as to whether a
particular larva should be classified as mandibulate, caudate, vesiculate, or
polypodeiform, for it may possess two, or in some cases three, of the
characters upon which the grouping is based.
The hymenopteriform larva has a medium‑sized head, 13 body
segments, which usually bear transverse bands or rows of setae, and spiracles
on the first thoracic and the first eight abdominal segments. This type of larva is representative of
the ectophagous forms, comprising the Vipioninae and representatives of the
Braconinae and Horminae, and will doubtless be found in other groups. Bracon sp., probably B. hylobii
Ratz., is distinguished by the lack of spiracles (Munro, 1917). The mandibulate larvae are found
generally in the Opiinae and, in combination with the caudate character, in
the Euphorinae, Triaspinae, Alysiinne, and Pambolinae. Vesicle‑bearing larvae of this type
occur in the Macrocentrinae. The
larva of Opius tryoni (Pemberton and Willard,
1918) is typical of the Opiinae and has a large, heavily sclerotized head,
large falcate mandibles, and short, blunt antennae. A pair of fleshy finger‑like
processes is found ventrally at the anterior margin of the first thoracic
segment. A well‑defined tracheal
system, with anterior and posterior commissures, is present and filled with
air, but there are no spiracles. The
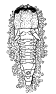
larvae of O. humilis, O. fletcheri (Fig.
15), and O. fullawayi are very similar to
that of O. tryoni. Keilin and Picado (l913) have made an
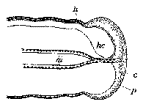
extended study of the first‑instar larva of O. crawfordi (Fig.
16A), which has an almost spherical head, a pair of very large mamma‑like
processes, each surmounted by three sensory papillae, on the first thoracic
segment and a smaller conical‑shaped pair on the third segment. These processes are on the concave side of
the body, as is also the anal opening.
The authors assert that the concave side of the body, to which the
mouth opening is directed, is in reality the dorsum and support their
conclusion by demonstrating the presence of the nerve cord along the convex
side and of the heart on the concave side. Recognition of the markedly
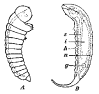
concave side of the first‑instar larva as dorsal rather than ventral is
also reported by Baume‑Pluvinel (1914) in Adelura gahani
B.‑P. (Fig. 16B), which
develops in the larvae of various Phytomyzinae. Further investigation of this interesting point in other
species would be desirable. The larva of Ascogaster quadridentata
has an exceedingly large head and 13 body segments of diminishing width, with
no tail or other fleshy processes, whereas Chelonus annulipes,
which is of modified mandibulate form, has a short tail following the 8 or
less distinct body segments. The
larva of Macrocentrus ancylivoruss is elongate in
form and has a pair of short fleshy processes and a short caudal horn on the
last segment. The anal vesicle is
relatively small. In the Euphorinae
the large, heavily sclerotized head, bearing the falcate mandibles at the
front, is followed by 12 or 13 body segments of decreasing width and a rather
short tail, which bears setae on the distal half or two‑thirds. The true vesticulate larvae are
found mainly in the subfamily Microgasterinae, of which the principal studies
have been made in the genera Apanteles
and Microgaster. At the time of hatching, many of these
have the general appearance of mandibulate larvae, and they may bear a fleshy
tail approaching half the length of the body proper. Usually only 10 or 11 ring-like body
segments are distinguishable, the last segment apparently representing
several that have fused. Each of the
segments usually bear a transverse row of setae dorsally. In A.
tasmanica Cam. (Dumbleton
1935) and Miscogaster tibialis (Fig. 17a) (Vance,
1932a), the rows of setae are lacking on the first two segments, while in
other species they are missing on only the first segment. The vesicle appears shortly after feeding
begins, and its width is then equal to or greater than that of the preceding
segments (Fig. 17C). The body at this
time is somewhat cylindrical, and the tail, which previously was prominent,
now appears as only a small ventrally directed "horn" beneath the
vesicle. In A. militaris
(Tower 1915), A. hyphantriae Riley, and A. thompsoni, there is no indication of a tail structure at
any time, and the bulb-like vesicle is well developed even before hatching. Please CLICK on
pictures to view details:
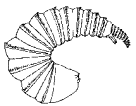
The wall of the proctodeum of the first‑instar larva of Orgilus obscurator Nees is relatively thin (Fig. 18), but it
increases greatly in thickness in the second instar (Thorpe 1932). The simple caudate type of larva,
without other adaptive modifications, is found principally in the Meteorinae
and Aphidiinae, whereas the tail in some form is present in practically all
groups which develop internally. In Meteorus, there are 12 or 13
segments exclusive of the tail, and the latter may exceed the body in length,
though in some species it is only one‑eighth as long. Each segment usually bears a transverse
row of setae on the dorsum, and the tail may also bear setae. The first‑instar larvae of many of
the Aphidiinae are recognizable principally by the possession of a row or
comb of heavy setae at the posterior margins of each body segment and by the
two ventrally directed lobes of the last segment. The tail in this subfamily is usually somewhat tubular in form
with the distal end rounded. There
are 13 distinct body segments. The
fringe of spines at the posterior margin of each segment dorsally and
extending to the lateroventral margins is, so far as known, found only in Praon, while in P. simulans, studied
by Timberlake (1910), they occur only on the third thoracic segment and on
all abdominal segments except the last.
Janiszewska (1933) describes the larva of an undetermined Aphidiine
believed to be Aphidius, in
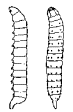
which this row or comb of spines is present on each body segment. In Ephedrus
incompletus Prov. (Wheeler
1923) and other species of the genus, the larva (Fig. 19) bears on each
segment a median transverse ridge which is more pronounced on the dorsum and
sides and is strongly serrate, with the teeth directed caudad. The tail also is heavily and completely
spined, with the spines arranged in rings about it. These adaptations are
possibly locomotory in function. The
ventrally directed bilobed process of the caudal segment is found in Praon, in Ephedrus, and in some species of Aphidius, and consists of two conical or finger‑like
processes, about the length of one segment, situated ventrally at the base of
the tail. The majority of the species
of the genus Aphidius have
simple caudate larvae, which lack entirely the integumentary spines and the
paired caudal process, and the tail is only lightly spined on its distal
portion. Larvae of the Aphidiinae
have the anal opening ventrally at the base of the tail rather than dorsally;
in the species having the lobed processes, it is situated between the bases
of the lobes and the tail. The polypodeiform larva is found
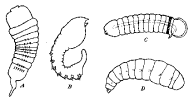
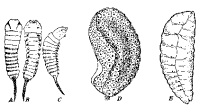
in isolated species in a number of subfamilies. That of Dacnusa
navicularis var. cynaraphila Ric. (Ricchello
1928) (Fig. 20A) is, except for its paired ventral processes, typically
caudate, with a transverse row of setae dorsally on each abdominal
segment. The paired ventral processes
occur upon each of the 12 body segments and are surmounted by a group or row
of setae. The larva of D. areolaris (Fig.
20D), on the other hand, lacks the tail and the paired ventral
processes (Haviland, 1922a). Bassus dimidiator
(Fig. 21) (Silvestri, 1923a), B.
pumilus Ratz. (Thorpe,
1933), B. stigmaterus Cress, and Macrocentrus gifuensis are distinguished by
having two pairs of fleshy processes on each segment. In M.
gifuensis, these are present
on the first 12 segments and are of uniform size, whereas in the first two
species named they are lacking on the first segment and are of slightly
greater size on the abdomen.
The respiratory system of first‑instar larvae of certain of the
endoparasitic species, such as the Opiinae, consists of the two lateral
trunks with branches at the various segments and an anterior dorsal and a
posterior ventral commissure. In a considerable
number of species, however, there is a complete lack of the tracheal system
in this instar.
n the great majority of species, the mandibles are simple, though
several exceptions occur. Those of Microbracon brevicornis are dentate on the
lower border, whereas in Bracon
tachardiae they are dentate,
the teeth being long and spine‑like, and in Heterospsilus cephi
(Hill and Smith, 1931) the main tooth is followed by five or six elongate
teeth in comb‑like arrangement along the inner edge. It will be noted that these departures
from the normal are in species which feed externally.
Intermediate‑ and Final‑instar Larvae. --The intermediate
larval instars of the ectoparasitic species do not differ in any essential
character from the first instar.
Among the internal parasites, the mandibulate‑type larva loses
the large, heavily sclerotized head, with its long falcate mandibles, at the
first molt, and in the caudate forms the tail is reduced in size with each
succeeding molt and is entirely lacking in the last instar. In some species having five instars, it
disappears after the second molt, and in Cosmophorus
henscheli it is entirely
absent in the second and following instars.
Many first‑instar Apanteles
larvae have a tail, or "caudal horn," situated beneath the vesicle,
which in some species disappears entirely at the first molt and in others persists
in reduced size in the second‑instar larva. In contrast to this, the anal vesicle of the larvae that
possess it increases in size with each molt but is absent in the final
instar. In A. thompsoni,
it is said to be present for only a short time after the second molt. The paired ventral processes on the last
segment of many aphidiine larvae do not persist beyond the first instar. Many species reveal an increasing number
of small teeth on the inner margin of the mandibles in the intermediate
instars. In Bracon tachardiae
Cam., the four teeth of the first instar are succeeded by five in the second
and third, whereas the mandibles of Microbracon
mellitor Say are simple in
all instars.
The paired ventral processes that occur on the bodies of first‑instar
polypodeiform larvae, such as those of some species of Bassus and of Macrocentrus
gifuensis, persist in much
reduced form in the second instar.
According to Parker, the larva of the latter species lacks mandibles
in this instar.
The mature larvae of the Braconidae are of normal form and have few
characters that distinguish them, aside from the tracheal system. In many species, the mandibles have minute
teeth, often slender and spine‑like, on the inner margin, approaching
30 in number in some species, giving a comblike appearance. Voukassovitch, in describing the mature
larva of M. abdominalis mentions a bilobed
chitinous "anal capsule," of which the ventral lobe is more heavily
sclerotized and bears a small ventrally directed process. The anal opening is between the two lobes
of the capsule. Beeson and Chatterjee
refer to a prominent "process" ventrally on the fifth and sixth
abdominal segments of the larva of Perilitus
mylloceri Wlkn. but do not
otherwise describe it. The mature larvae of many of the
ectophagous species bear a dense coating of fine hairs; in some instances,
this is uniform over the body, and in others it occurs in a transverse band
on each segment and may be absent ventrally. It has already been pointed out
that the respiratory system of the ectoparasitic first‑instar braconid
larva has normally nine pairs of spiracles, situated on the first thoracic
and the first eight abdominal segments In these species, largely included in
the Vipioninae, this number and arrangement persist through all the following
instar. The early‑instar larvae
of the species that develop internally lack the open tracheal system; and, in
species that are known to have five instars, the spiracles first appear on
the fourth. The species in which only
three or four larval instars have been distinguished reveal the spiracles
only on the last instar. De Leon (1934) has summarized the
information available regarding the respiratory system of mature braconid
larvae and has attempted to group the subfamilies on the basis of spiracle
number and position and on the presence or absence of certain
commissures. The information
available is sufficient for only a very few subfamilies to permit of
generalizations in this respect. It
appears, however, that the Vipioninae quite consistently have the number and
arrangement given above, and limited information indicates that this is true
of the Braconinae also. The most
common spiracular arrangement, however, has the same number, but the thoracic
pair is situated on the second segment rather than the first. This order appears to predominate in the
Macrocentrinae, Meteorinae, Euphorinae, Opiinae, and Alysiinae. Macrocentrus
ancylivorus is said to have
the spiracles on the second and third thoracic and the second to eighth
abdominal segments, whereas M.
abdominalis has 10 pairs,
the additional one being upon the ninth abdominal segment. In the Microgasterinae, the occurrence of
eight pairs is quite general, and in most species the single thoracic pair is
on the second segment, whereas in a smaller number it is upon the third. Microgaster
connexus, however, has only
six abdominal pairs rather than seven, and Apanteles lictorius
Rein. is said to have nine pairs,
though their position is not given. On the basis of information
regarding a limited number of species, it seems that the greatest variation
in spiracle arrangement occurs in the Aphidiinae. Aphidius granarius L. has spiracles on
the first thoracic and eight abdominal segments, and Ephedrus plagiator
Nees (Skriptshinskij, 1930) on the second and third thoracic and seven
abdominal segments. Wheeler (1923)
states that tracheal system and spiracles are absent in the aphidiine species
studied, representing three genera but this is so unusual as to require
verification. The tracheal system of the mature
braconid larvae is distinguished from that of the Ichneumonidae chiefly by
the absence of the secondary lateral commissures in the thorax, which connect
with the main trunks by three branches.
The anterior dorsal commissure is present in all species, but the
absence of the posterior ventral commissure has been noted in species of Chelonus, Apanteles, Microplitis,
and Meteorus. Ventral abdominal commissures occur in the
first eight segments in several species of Vipioninae and in Doryctes gallicus Rh. Among the species that develop
within the host, many have an internal tracheal system but no spiracles in
the early instars, and the spiracles appear only in the last larval
instar. In Bracon sp. (probably B. hylobii) studied by Munro (1917),
which is the single species of external habit that lacks spiracles in the
early instars, they appear first in the fourth (penultimate) instar. According to Glover (1934), who
has studied the immature forms of B.
tachardiae, the head widths
of the five larval instars conform to Dyar's principle, though the extremes
overlap, whereas mandible length shows no overlapping. De Stephani Perez (1902) has
described the so‑called chrysalis of Giardinaia urinator
Perez, found upon the stems of Potamogeton,
which he considers to be the last larval exuviae and within which pupation is
said to take place. The last body
segment is bifurcate, and the terminal "hooks" are embedded in the
stem of the plant. They may bear
spiracles, and the supply of air would thus be derived from the plant. The figure of this chrysalis shows 14
segments. The parasite pupa
illustrated within it is slightly more than half its length and one‑sixth
to one‑eighth its volume. At
emergence, the wasp breaks through the dorsum of the chrysalis and climbs to
the surface of the water. It is
extremely improbable that this chrysalis is the last larval skin of Giardinaia; it is much more
likely to be that of its host. This
parasite has been recorded from Hydrellia
sp. in Europe and the "chrysalis" described by Perez may be the
empty larval skin of a species of Ephydridae. The aquatic larvae of some representatives of this family are
known to possess caudal spiracles upon a bifurcate process and to derive
their air supply from plants (Clausen 1940). References: Please refer to <biology.ref.htm>, [Additional references may
be found at: MELVYL Library ] |